Physics-
General
Easy
Question
A cyclic process ABCA is shown in PT diagram. When presented on PV, it would

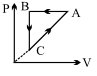

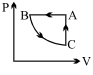

The correct answer is: 
Related Questions to study
physics-
An ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?

An ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?

physics-General
physics-
A closed container is fully insulated from outside. One half of it is filled with an ideal gas X separated by a plate P from the other half Y which contains a vacuum as shown in figure. When P is removed, X moves into Y. Which of the following statements is correct?

A closed container is fully insulated from outside. One half of it is filled with an ideal gas X separated by a plate P from the other half Y which contains a vacuum as shown in figure. When P is removed, X moves into Y. Which of the following statements is correct?

physics-General
physics-
Three processes compose a thermodynamics cycle shown in the PV diagram. Process  takes place at constant temperature. Process
takes place at constant temperature. Process  takes place at constant volume, and process
takes place at constant volume, and process  is adiabatic. During the complete cycle, the total amount of work done is 10 J. During process
is adiabatic. During the complete cycle, the total amount of work done is 10 J. During process  , the internal energy decrease by 20J and during process
, the internal energy decrease by 20J and during process  , 20 J of work is done on the system. How much heat is added to the system during process
, 20 J of work is done on the system. How much heat is added to the system during process  ?
?

Three processes compose a thermodynamics cycle shown in the PV diagram. Process  takes place at constant temperature. Process
takes place at constant temperature. Process  takes place at constant volume, and process
takes place at constant volume, and process  is adiabatic. During the complete cycle, the total amount of work done is 10 J. During process
is adiabatic. During the complete cycle, the total amount of work done is 10 J. During process  , the internal energy decrease by 20J and during process
, the internal energy decrease by 20J and during process  , 20 J of work is done on the system. How much heat is added to the system during process
, 20 J of work is done on the system. How much heat is added to the system during process  ?
?

physics-General
physics-
Two identical vessels A & B contain equal amount of ideal monoatomic gas. The piston of A is fixed but that of B is free. Same amount of heat is absorbed by A & B. If B's internal energy increases by 100 J the change in internal energy of A is

Two identical vessels A & B contain equal amount of ideal monoatomic gas. The piston of A is fixed but that of B is free. Same amount of heat is absorbed by A & B. If B's internal energy increases by 100 J the change in internal energy of A is

physics-General
physics-
A gas takes part in two processes in which it is heated from the same initial state 1 to the same final temperature. The processes are shown on the P-V diagram by the straight line 1-2 and 1-3. 2 and 3 are the points on the same isothermal curve. Q1 and Q2 are the heat transfer along the two processes. Then

A gas takes part in two processes in which it is heated from the same initial state 1 to the same final temperature. The processes are shown on the P-V diagram by the straight line 1-2 and 1-3. 2 and 3 are the points on the same isothermal curve. Q1 and Q2 are the heat transfer along the two processes. Then

physics-General
physics-
A ideal monoatomic gas is carried around the cycle ABCDA as shown in the fig. The efficiency of the gas cycle is

A ideal monoatomic gas is carried around the cycle ABCDA as shown in the fig. The efficiency of the gas cycle is

physics-General
physics-
Three processes form a thermodynamic cycle as shown on P-V diagram for an ideal gas Process 1 → 2 takes place at constant temperature (300K).Process  takes place at constant volume During this process 40J of heat leaves the system Process
takes place at constant volume During this process 40J of heat leaves the system Process  is adiabatic and temperature T3 is 275K. Work done by the gas during the process
is adiabatic and temperature T3 is 275K. Work done by the gas during the process
 is
is

Three processes form a thermodynamic cycle as shown on P-V diagram for an ideal gas Process 1 → 2 takes place at constant temperature (300K).Process  takes place at constant volume During this process 40J of heat leaves the system Process
takes place at constant volume During this process 40J of heat leaves the system Process  is adiabatic and temperature T3 is 275K. Work done by the gas during the process
is adiabatic and temperature T3 is 275K. Work done by the gas during the process
 is
is

physics-General
physics-
A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is 3 times the amount of work done by the cycle, the efficiency of the cycle is
A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is 3 times the amount of work done by the cycle, the efficiency of the cycle is
physics-General
physics-
The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases A and B undergoing adiabatic process. From figure it can be concluded that

The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases A and B undergoing adiabatic process. From figure it can be concluded that

physics-General
physics-
An ideal gas is taken from point A to point C on P-V diagram through two process AOC and ABC as shown in the figure. Process AOC is isothermal

An ideal gas is taken from point A to point C on P-V diagram through two process AOC and ABC as shown in the figure. Process AOC is isothermal

physics-General
physics-
Pressure versus temperature graph of an ideal gas is shown in figure

Pressure versus temperature graph of an ideal gas is shown in figure

physics-General
physics-
In the above thermodynamic process, the correct statement is

In the above thermodynamic process, the correct statement is

physics-General
physics-
An ideal gas undergoes the process 1  2 as shown in the figure, the heat supplied and work done in the process is
2 as shown in the figure, the heat supplied and work done in the process is  and
and  respectively. The ratio
respectively. The ratio  is
is

An ideal gas undergoes the process 1  2 as shown in the figure, the heat supplied and work done in the process is
2 as shown in the figure, the heat supplied and work done in the process is  and
and  respectively. The ratio
respectively. The ratio  is
is

physics-General
physics-
A given mass of a gas expands from a state A to the state B by three paths 1, 2 and 3 as shown in T-V indicator diagram. If W1, W2 and W3 respectively be the work done by the gas along the three paths, then

A given mass of a gas expands from a state A to the state B by three paths 1, 2 and 3 as shown in T-V indicator diagram. If W1, W2 and W3 respectively be the work done by the gas along the three paths, then

physics-General
physics-
At temperature T, N molecules of gas A each having mass m and at the same temperature 2N molecules of gas B each having mass 2m are filled in a container. The mean sqaure velocity of molecules of gas B is v2 and mean square of x component of velocity of molecules of gas A is w2. The ratio of w2/v2 is :
At temperature T, N molecules of gas A each having mass m and at the same temperature 2N molecules of gas B each having mass 2m are filled in a container. The mean sqaure velocity of molecules of gas B is v2 and mean square of x component of velocity of molecules of gas A is w2. The ratio of w2/v2 is :
physics-General





