Maths-
General
Easy
Question
Alex went to market to purchase tomatoes. He bought 4.5Kg tomatoes at the rate of $12 per kg. How much did he spent ?
- $45
- $30
- $54
- $25
The correct answer is: $54
Cost of 1kg tomatoes = $12
Cost of 4.5kg = 4.5 x 12 = $54
Related Questions to study
Maths-
The dimensions of room are 8m , 12 m and 10m. Find the lengths of the measuring tape which can measure it in equal parts.
The dimensions of room are 8m , 12 m and 10m. Find the lengths of the measuring tape which can measure it in equal parts.
Maths-General
Maths-
Out of 150 marks, Alisha scored 120. What is her percentage?
Out of 150 marks, Alisha scored 120. What is her percentage?
Maths-General
Maths-
“All similar triangles are congruent”
“All similar triangles are congruent”
Maths-General
Maths-
Joey scored 75, 80,76,85 and 90 in five subjects out 100 each. Calculate his percentage?
Joey scored 75, 80,76,85 and 90 in five subjects out 100 each. Calculate his percentage?
Maths-General
Maths-
Find the number by which 7:21 is multiplied to make it equivalent to 84 : 252.
Find the number by which 7:21 is multiplied to make it equivalent to 84 : 252.
Maths-General
Chemistry-
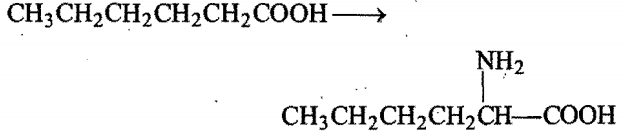 the reagents used in the conversion are:
the reagents used in the conversion are:
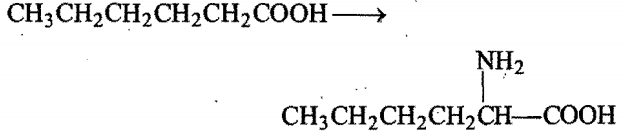 the reagents used in the conversion are:
the reagents used in the conversion are:
Chemistry-General
Chemistry-
Which is the correct order of acidity from weakest to strongest acid for these compounds:
I) CF3CH2OH
II) 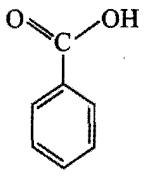
III) 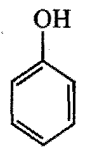
IV) 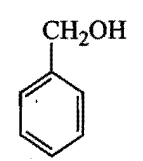
Which is the correct order of acidity from weakest to strongest acid for these compounds:
I) CF3CH2OH
II) 
III) 
IV) 
Chemistry-General
Chemistry-
Two aliphatic aldehydes P and Q react in the presence of aqueous K2CO3 to give compound R, which upon treatment with HCN provides compound S On acidification and heating S gives the product shown below:
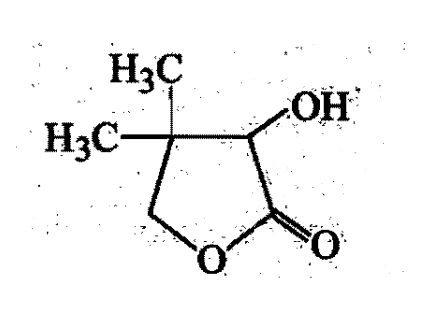
The compound P and Q respectively are :
Two aliphatic aldehydes P and Q react in the presence of aqueous K2CO3 to give compound R, which upon treatment with HCN provides compound S On acidification and heating S gives the product shown below:

The compound P and Q respectively are :
Chemistry-General
Chemistry-
A tertiary alcohol H on acid catalysed dehydration gives a product I. Ozonolysis of I leads to compounds J and K. Compound J upon reaction with KOH gives benzyl alcohol and a compound L, whereas K on reaction with KOH gives only M
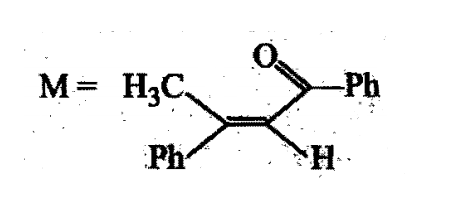
The structures of compounds J, K and L, respectively, are:
A tertiary alcohol H on acid catalysed dehydration gives a product I. Ozonolysis of I leads to compounds J and K. Compound J upon reaction with KOH gives benzyl alcohol and a compound L, whereas K on reaction with KOH gives only M

The structures of compounds J, K and L, respectively, are:
Chemistry-General
Chemistry-
A tertiary alcohol H on acid catalysed dehydration gives a product I. Ozonolysis of I leads to compounds J and K. Compound J upon reaction with KOH gives benzyl alcohol and a compound L, whereas K on reaction with KOH gives only M
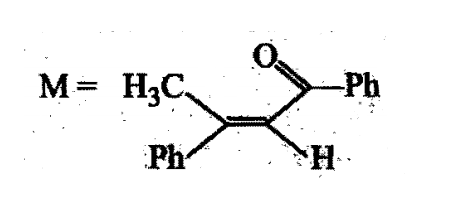
The structure of compound I is:
A tertiary alcohol H on acid catalysed dehydration gives a product I. Ozonolysis of I leads to compounds J and K. Compound J upon reaction with KOH gives benzyl alcohol and a compound L, whereas K on reaction with KOH gives only M

The structure of compound I is:
Chemistry-General
Chemistry-
A tertiary alcohol H on acid catalysed dehydration gives a product I. Ozonolysis of I leads to compounds J and K. Compound J upon reaction with KOH gives benzyl alcohol and a compound L, whereas K on reaction with KOH gives only M
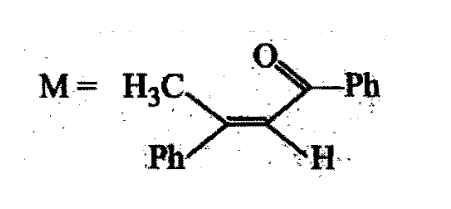
Compound H is formed by the reaction of:
A tertiary alcohol H on acid catalysed dehydration gives a product I. Ozonolysis of I leads to compounds J and K. Compound J upon reaction with KOH gives benzyl alcohol and a compound L, whereas K on reaction with KOH gives only M

Compound H is formed by the reaction of:
Chemistry-General
Chemistry-
Aldehydes undergo disproportionation reaction in presence of aqueous NaOH Simultaneous oxidation and. reduction 'of a compound is scientifically called as disproportionation.
Aldehydes having no a-hydrogen show this reaction called Cannizzaro's reaction. Few exceptions are also there to this generalization.
The reaction may be represented as:

Intramolecular Cannizzaro's reaction is also possible.
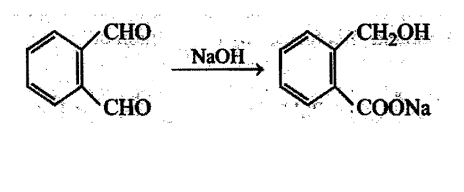
Which of the following' compounds gives internal crossed Cannizzaro's reaction?
Aldehydes undergo disproportionation reaction in presence of aqueous NaOH Simultaneous oxidation and. reduction 'of a compound is scientifically called as disproportionation.
Aldehydes having no a-hydrogen show this reaction called Cannizzaro's reaction. Few exceptions are also there to this generalization.
The reaction may be represented as:

Intramolecular Cannizzaro's reaction is also possible.

Which of the following' compounds gives internal crossed Cannizzaro's reaction?
Chemistry-General
Chemistry-
Aldehydes undergo disproportionation reaction in presence of aqueous NaOH Simultaneous oxidation and. reduction 'of a compound is scientifically called as disproportionation.
Aldehydes having no a-hydrogen show this reaction called Cannizzaro's reaction. Few exceptions are also there to this generalization.
The reaction may be represented as:

Intramolecular Cannizzaro's reaction is also possible.
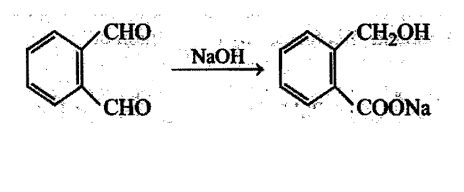
The aldehyde having  -hydrogen which gives Cannizzaro's reaction is:
-hydrogen which gives Cannizzaro's reaction is:
Aldehydes undergo disproportionation reaction in presence of aqueous NaOH Simultaneous oxidation and. reduction 'of a compound is scientifically called as disproportionation.
Aldehydes having no a-hydrogen show this reaction called Cannizzaro's reaction. Few exceptions are also there to this generalization.
The reaction may be represented as:

Intramolecular Cannizzaro's reaction is also possible.

The aldehyde having  -hydrogen which gives Cannizzaro's reaction is:
-hydrogen which gives Cannizzaro's reaction is:
Chemistry-General
Chemistry-
Aldehydes undergo disproportionation reaction in presence of aqueous NaOH Simultaneous oxidation and. reduction 'of a compound is scientifically called as disproportionation.
Aldehydes having no a-hydrogen show this reaction called Cannizzaro's reaction. Few exceptions are also there to this generalization.
The reaction may be represented as:

Intramolecular Cannizzaro's reaction is also possible.
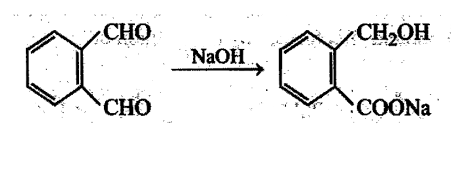
The aldehyde which shows Cannizzaro's reaction is:
Aldehydes undergo disproportionation reaction in presence of aqueous NaOH Simultaneous oxidation and. reduction 'of a compound is scientifically called as disproportionation.
Aldehydes having no a-hydrogen show this reaction called Cannizzaro's reaction. Few exceptions are also there to this generalization.
The reaction may be represented as:

Intramolecular Cannizzaro's reaction is also possible.

The aldehyde which shows Cannizzaro's reaction is:
Chemistry-General
Chemistry-
The final product (III) obtained in the reaction, 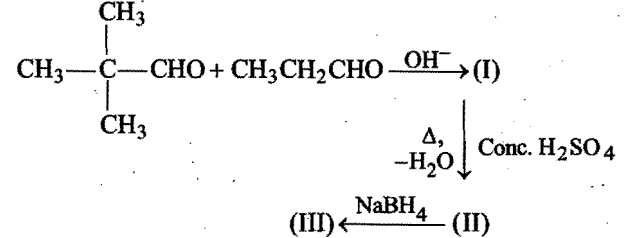 is:
is:
The final product (III) obtained in the reaction, 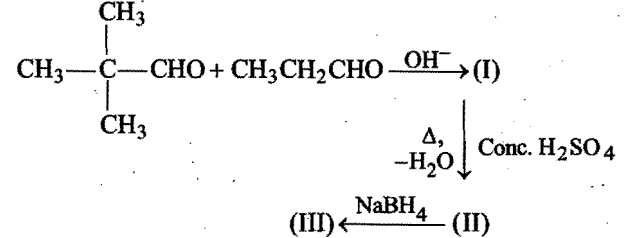 is:
is:
Chemistry-General



